LIDOCAINE TO REDUCE THE SEVERITY OF COVID-19 CASES
Maria Ayeza Diaz-Vera*, José Terrones Santa Cruz**, Aldo Forttini Headrington***, Javier A Cerna Paz***, Luis Quintanilla Rios***, Moises P Medina Melendez***, José Del Aguila Torres****
*Pediatrician, graduated in Alternative and Complementary Therapy; **Emergency Medicine Physician
***Medical Specialist in Internal Medicine; ****Medical specialist in Anesthesiology
San Juan de Lurigancho, Lima, Perú
October 2020
Versión Español · See PDF Free
*Pediatrician, graduated in Alternative and Complementary Therapy; **Emergency Medicine Physician
***Medical Specialist in Internal Medicine; ****Medical specialist in Anesthesiology
San Juan de Lurigancho, Lima, Perú
October 2020
Versión Español · See PDF Free
Citation: Diaz-Vera MA, Terrones Santa Cruz J, Forttini Headrington A, Cerna Paz JA, Quintanilla Rios L, Medina Melendez MP, Del Aguila Torres J. Lidocaine to reduce the severity of COVID-19 cases [Internet]. Sabadell, Barcelona (Spain); Publicated Nov 19th 2020. Avaiable in : www.terapianeural.com
SUMMARY
Yang et al. have shown through a meta-analysis that includes 4,292 articles that low-dose lidocaine is capable of reducing cough for endotracheal intubation and significantly reduces sore throat (11). Aminajad et al. observe the same symptom-reducing effect in patients with Covid 19 (18). Likewise, there is a large number of studies that show that local anesthetics, among them the most widely used, Lidocaine, have important effects that prevent cellular damage produced by inflammation in the lung and other organs (6,7,8,9, 10.21).
On the other hand, multiple studies that show that the severe and moderate effects of the SARS-Cov2 coronavirus in humans are due to a dysregulated inflammation, that is, a great tissue immune response, especially in the lung, which produces pneumonia, heart attacks, kidney damage, etc., this response is known as the cytokine storm (1,2,3) and reaches the Hemophagocytic syndrome (4,5) and disseminated intravascular coagulation, this tissue damage leads to multi-organ dysfunction syndrome and death.
Therefore, it can be inferred that Lidocaine could attenuate the symptoms of Covid 19, a clinical trial was carried out, administering intravenous Lidocaine at doses of 0.5 to 1 mg/kp per day, the dose used in various studies (11), to attenuate the symptoms caused by SARS-Cov2, hoping to reduce the severity of the symptoms produced by the new coronavirus that has been affecting the world population.
INTRODUCTION
The coronavirus 2 produces severe acute respiratory syndrome in English acronym: SARS-CoV-2 is a new type of coronavirus that causes the disease called by the WHO as "coronavirus disease of 2019" is abbreviated by its acronym in English COVID- 19. It was discovered and isolated for the first time in Wuhan, China, after causing the 2019-2020 coronavirus disease pandemic. (14)
The effects of SARS-Cov2 in humans can be divided into mild, moderate, and severe. The severity of the symptoms is directly related to the type of tissue immune response that it generates, being in some people very severe, especially in the lung, which produces pneumonia, besides heart attacks, kidney damage, etc., tissue damage reaches syndrome of multiorgan dysfunction and death, this response is known as the cytokine storm (1,2,3) and progresses to Hemophagocytic syndrome (4,5), in the current pandemic, based on animal studies and autopsies of the deceased, this type of immune response results in severe cases and mortality.
The two previously reported coronavirus epidemics (severe acute respiratory syndrome known by its acronym SARS and Middle East respiratory syndrome [MERS]) share pathogenic, epidemiological, and clinical features of COVID-19. Little is currently known about SARS-CoV-2, and it is likely that the lessons learned from these large epidemics can be applied to the new pandemic, including the use of new immunosuppressive drugs. (3)
EFFECT OF LOCAL ANESTHETICS ON THE INFLAMMATION PROCESS
Studies of humans who died of SARS and more recent studies in animal models suggested that a dysregulated immune response occurs in COVID-19 cases, resulting in exuberant inflammation and fatal disease (1). Local anesthetics have been shown to be protective against SARS in various animal models and the underlying mechanism appears to be their anti-inflammatory action. (15)
Recently, different properties of lidocaine have been found, some of which exceed its characteristics as a local anesthetic. These properties will depend on the different plasma concentrations at which it is found. Effects on sodium channels occur at 50-100 µM; However, at lower concentrations, different effects have been attributed to them such as neuroprotection, antithrombotics, platelet aggregation, as an antiarrhythmic, wound healing, tinnitus remission, bronchial hyperresponsiveness as an antinociceptive, as a muscarinic blocker and dopamine and finally anti-inflammatory properties at 0.5-5.0 μg / mL or 2-20 μM (7).
Lidocaine has several inhibitory effects on neutrophil function. Reduces adhesion of leukocytes to microvascular endothelium and non-biological surfaces. It also inhibits leukocyte migration to the inflammatory site. Furthermore, research has shown that lidocaine attenuates the white blood cell production of prostaglandins, leukotrienes, and thromboxanes. Lidocaine also reduces the production of free radicals by neutrophils and inhibits the release of superoxide anions. Furthermore, it decreases the release of protease from neutrophils and macrophages by stabilizing their cell membranes. (6)
Hollman (15) argues that the actions of local anesthetics are very promising for clinical application: their effects on the inflammatory response and especially on inflammatory cells (mainly polymorphonuclear granulocytes [PMNs] but also macrophages and monocytes). Hyperactive inflammatory responses that destroy rather than protect are critical in the development of various perioperative disease states, such as postoperative pain, adult respiratory distress syndrome (ARDS), systemic inflammatory response syndrome, and multiple organ failure. Perioperative modulation of such responses is therefore relevant to the practice of anesthesiology, and local anesthetics may play a significant role in this regard.
Ochoa (7) in his review of the properties of Lidocaine analyzes various studies carried out in animals where positive effects of this local anesthetic in SARS are observed. Such as the study by Nishina (16) in which Lidocaine attenuates lung injury in animals, as well as the work by Mikawa (17). In the same way, De Klaver (10) concludes that intravenous lidocaine reduces endothelial injury produced by cytokines, likewise, lidocaine can also be used as a preventive of endotoxic lesions. (10.17)
Aminnejad (18) likewise Pandey (19) recommend an intravenous dose of Lidocaine before intubating patients with COVID-19 because they report that it is sufficient to suppress the cough. They are used during intubation and extubation of Covid-19 patients. Therefore, it is deduced that the patient improves clinically with a single dose of IV Lidocaine of 0.5 mg/kp.
Similarly, Yang et al. (11) Performed a meta-analysis in which they evaluated 4,293 articles in the main databases of scientific information and concluded that the use of intravenous lidocaine, compared with placebo, led to a large reduction in the incidence of post-cough. extubation with moderate quality of evidence. This large effect had an indicator Number Needed to Treat (NNT) of five, suggesting that its use would potentially benefit numerous surgical patients if it were to be implemented in clinical practice. Therefore, the evidence from this meta-analysis was sufficient to conclude the effectiveness of intravenous lidocaine in preventing cough at intubation, and it is unlikely that further trials will be conducted to alter this conclusion. The average dose used in the studies mentioned in the meta-analysis by Yang et al. they were between 1 to 1.5 mg/kp.
Ochoa (7) concludes that lidocaine is one of the most easily accessible drugs and that at low doses it can contribute to attenuate the inflammation produced by the cytokine storm and we can add that it could reduce the damage of the new coronavirus to the pandemic that is affecting to the world population. On the other hand, Mitra (20) reports that the administration of Lidocaine could also be beneficial for the side effects of hydroxychloroquine in QT.
METHODOLOGY
Lidocaine without epinephrine and without preservatives was administered to patients diagnosed with COVID 19 at doses of 0.5 to 1 mg/kp intravenously diluted as 0.5% lidocaine, once a day for 2 days and the following parameters were evaluated: pain, cough, respiratory rate, saturation. In some outpatient cases, undiluted 2% lidocaine was administered subcutaneously at a dose of 1 mg/kp. Pain was evaluated using the Visual Analogue Scale (VAS).
RESULTS
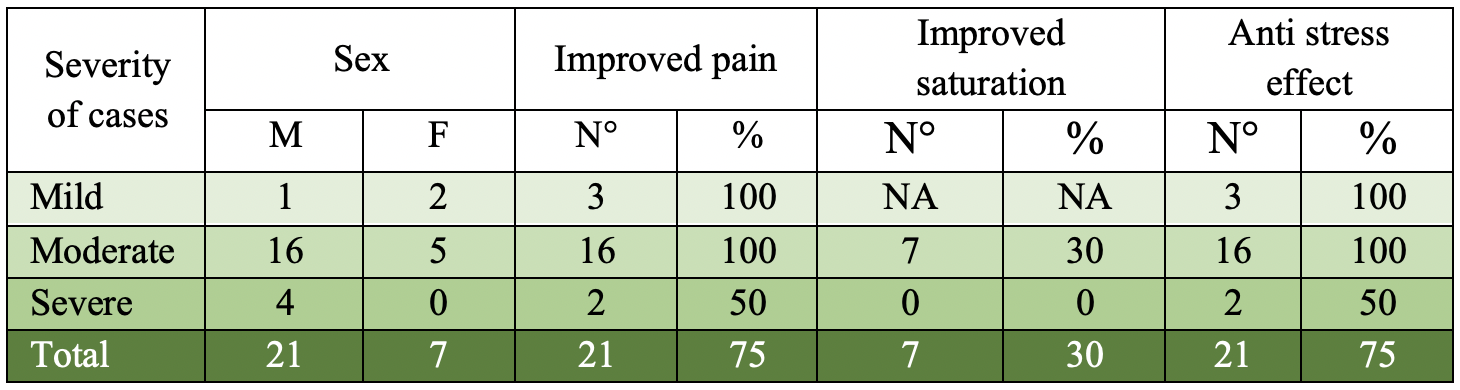
28 patients with a diagnosis of Covid 19 were evaluated, between 28 and 85 years old, 70% were male, 70% of moderate cases were found whose main symptoms were respiratory distress with hypoxia and in most the pain was intense, mainly manifested such as headaches, chest pain, and low back pain that did not improve with standard medications, which often prevent patients from functions such as eating and/or sleeping. It was found that 100% Mild cases referred to a decrease in pain and discomfort at the time of evaluation, the patient felt calmer and more relaxed, and managed to fall asleep. In moderate cases, at the time of evaluation, it was found, in addition to a decrease in pain, a feeling of tranquility and sleep, an improvement in oxygen saturation, and a decrease in respiratory rate, in 30% of cases, that it remained at least 24 hours. In severe cases, the improvement of pain and general condition takes a little longer, but a feeling of tranquility was observed in patients, although saturation does not improve, stress decreases and in most patients it allows them to sleep. It decreases the cough, we could observe that in severe cases the medication calms the symptoms for shorter periods, between 3 to 4 hours, so we suppose they need more doses or the administration in infusion as is the usual use of lidocaine in ICU.
RECOMMENDATIONS
- Lidocaine attenuates the symptoms of COVID 19, reducing pain in a lasting way.
- In moderate cases of short evolution, saturation improves, and respiratory rate decreases, reducing the need for hospitalization in some patients.
- In severe cases stabilizes the patient, pain relief is slower, stress is reduced.
- In all cases there is a feeling of well-being, relaxation, can produce sleep, allows you to eat and since biological functions are frequently altered by discomfort, pain, and shortness of breath, Lidocaine improves the general condition,
- Improves user mood and satisfaction, especially with intravenous use.
- More studies are needed to evaluate the possibility of a decrease in the mortality rate, antiviral effects, and/or anti-inflammatory effects.
REFERENCES
- Channappanavar, R., Perlman, S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Revista Seminars in Immunopathol 39, 529–539 (2017). https://doi.org/10.1007/s00281-017-0629-x Disponible en: https://pubmed.ncbi.nlm.nih.gov/28466096/
- Sarzi-Puttini P, Giorgi V, Sirotti S, Marotto D, Ardizzone S, Rizzardini G, Antinori S, Galli M, COVID-19, cytokines and immunosuppression: what can we learn from severe acute respiratory syndrome? Revista Clinical and Experimental Rheumatology 2020;38(2):337-42. Disponible en: https://www.clinexprheumatol.org/article.asp?a=15518
- Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16–32. doi:10.1128/MMBR.05015-11. Disponible en: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3294426/
- Mehta P, McAuley DF, Brown M, Sánchez E, Tattersall RS, Manson JJ, et al, COVID-19: consider cytokine storm syndromes and immunossuppresion, The Lancet 2020;395:1033-34, Correspondense. Disponible en: https://www.thelancet.com/action/showPdf?pii=S0140-6736%2820%2930628-0
- Rouphael Maakaroun N, Moanna A, Jacob JT, Albrecht H, Viral infections associated with haemophagocytic syndromes Rev. Med. Virol. 2010; 20: 93–105. Disponible en: https://pubmed.ncbi.nlm.nih.gov/20127750/ 1034, MARCH 28, 2020
- Schmidt W, Schmidt H, Bauer H, Gebhard MM, Martín E, Influence od Lidocaine on Endotoxin-induced Leukocyte-Endothelial Cell Adhesion and Macromolecular Leakage in Vivo. Revista Anesthesiology 1997;87:617-24. American Society of Anathesiologists. Disponible en: https://anesthesiology.pubs.asahq.org/article.aspx?articleid=1947880
- Ochoa-Anaya G, Aguirre-Ibarra CP, Franco-Cabrera M. Lidocaína: aspectos generales y nuevas implicaciones en la inflamación. Artículo de Revisión, Rev Mex Anest. 2017;40(3):220-225. Disponible en: https://www.medigraphic.com/pdfs/rma/cma-2017/cma173j.pdf
- Griffin MJ, Letson HL, Dobson GP, Small-Volume Adenosine, Lidocaine, and Mg2 4-Hour Infusion Leads to 88% Survival after 6 Days of Experimental Sepsis in the Rat without Antibiotics, Revista Clinical and Vaccine Immunology 2016; Disponible en: https://pubmed.ncbi.nlm.nih.gov/27581435/
- Liu J, Zhang H, Qi Z, Zheng X, Lidocaine protects against renal and hepatic dysfunction in septic rats via downregulation of Toll‑like receptor 4, Revista Molecular Medicine Reports enero 2014; Volume 9 Issue 1, Published online on: November 14, 2013 https://doi.org/10.3892/mmr.2013.1799. Disponible en: https://www.spandidos-publications.com/mmr/9/1/118?text=fulltext
- De Klaver MJ, Buckingham MG, and Rich GF, Lidocaine Attenuates Cytokine-Induced Cell Injury in Endothelial and Vascular Smooth Muscle Cells Anesth Analg 2003;97:465–70. Disponible en: https://europepmc.org/article/med/12873936
- Yang SS, Wang NN, Postonogova T, Yang GJ, McGillion M, Beique F, Schricker T, Intravenous lidocaine to prevent postoperative airway complications in adults: a systematic review and meta-analysis. British Journal of Anaesthesia, 124 (3): 314e323 (2020). Disponible en https://bjanaesthesia.org/article/S0007-0912(19)30971-7/pdf
- El Confidencial, Mundo, El Coronavirus Supera las 100,000 muertes en todo el mundo, Diario El Confidencial, 10 de abril del 2020, España, https://www.elconfidencial.com/mundo/2020-04-10/el-coronavirus-supera-las-100-000-muertes-en-todo-el-mundo_2543884/
- Reuters, Economía mundial está en recesión por la devastación causada por el virus, aseguran expertos, América-Economía, 20 de marzo de 2020, Perú. Disponible en: https://www.americaeconomia.com/economia-mercados/finanzas/economia-mundial-esta-en-recesion-por-la-devastacion-causada-por-el-virus
- Centros para el Control y la Prevención de Enfermedades (CDC) «Enfermedad del coronavirus 2019 (COVID-19). Respuestas a las preguntas más frecuentes». Centros para el Control y Prevención de Enfermedades de los Estados Unidos de América. Consultado el 13 de abril de 2020.https://web.archive.org/web/20200318025404/https://www.cdc.gov/coronavirus/2019-ncov/faq-sp.html
- Hollmann MW, Durieux ME, Local Anesthetics and the Inflammatory Response. A New Therapeutic Indication? Revista Anesthesiology 2000; 93:858–75. American Society of Anesthesiologists USA. Disponible en: https://anesthesiology.pubs.asahq.org/article.aspx?articleid=1991071
- Nishina K, Mikawa K, Takao Y, Shiga M, Maekawa N, Obara H, Intravenous Lidocaine Attenuates Acute Lung Injury Induced by Hydrochloric Acid Aspiration in Rabbits. Revista Anesthesiology. 88(5):1300–1309, mayo 1998. Disponible en: https://anesthesiology.pubs.asahq.org/article.aspx?articleid=1947363
- Mikawa K, Maekawa N, Nishina K, Takao Y, Yaku H, Obara H, Effect of Lidocaine Pretreatment on Endotoxin-induced Lung Injury in Rabbits Anesthesiology. 81(3):689-699, september 1994. Disponible en: https://anesthesiology.pubs.asahq.org/article.aspx?articleid=1949463
- Aminnejad, R., Salimi, A. & Saeidi, M. Lidocaine during intubation and extubation in patients with coronavirus disease (COVID-19). Can J Anesth/J Can Anesth (2020). https://doi.org/10.1007/s12630-020-01627-2. Disponible en https://link.springer.com/article/10.1007/s12630-020-01627-2
- Pandey CK, Raza M, Ranjan R, et al. Intravenous lidocaine 0.5 mg.kg−1 effectively suppresses fentanyl-induced cough. Can J Anesth 2005; 52: 172-5. Disponible en: https://pubmed.ncbi.nlm.nih.gov/15684258/
- Mitra RL, Greenstein SA, Epstein LM, An algorithm for managing QT prolongation in Coronavirus Disease 2019 (COVID-19) patients treated with either chloroquine or hydroxychloroquine in conjunction with azithromycin: Possible benefits of intravenous lidocaine, HeartRhythm Case Reports (2020), doi: https://doi.org/10.1016/j.hrcr.2020.03.016. Disponible en: https://www.heartrhythmcasereports.com/article/S2214-0271(20)30057-9/abstract
- Colegio Mexicano de Medicina Crítica, Guía para la atención del Paciente Crítico con COVID-19. Marzo 2020. Disponible en: https://www.flasog.org/static/COVID-19/11_Abril_20_Final_compressed.pdf
